Iván M. Zerón, José Manuel Míguez, Jesús Algaba, Bruno Mendiboure, Felipe J. Blas, J. Colloid Interface Sci., 2025
https://doi.org/10.1016/j.jcis.2025.139477
Hypothesis: The interfacial free energy between hydrate and water phases is a key thermodynamic parameter that governs both nucleation kinetics and crystal growth of gas hydrates. In these strategic materials—crystalline inclusion compounds where hydrogen-bonded water cages encapsulate small guest molecules such as methane () and carbon dioxide ()—this interfacial energy plays a crucial role in determining phase stability and formation pathways. Given the significance of gas hydrates in energy storage, sequestration, and climate-related processes, accurately determining their interfacial energies is essential for advancing both fundamental understanding and technological applications. Despite its importance, the hydrate–water interfacial energy remains poorly constrained due to substantial experimental uncertainties and the limitations of indirect estimation methods. For example, reported experimental values for hydrate span a wide range from 28 to 40 mJ/m2 . Interestingly, some studies suggest these values are comparable to the interfacial free energy of the hexagonal ice (ice Ih)–water interface, approximately 32mJ/m2, hinting at potential analogies between clathrate hydrate and ice interfaces.
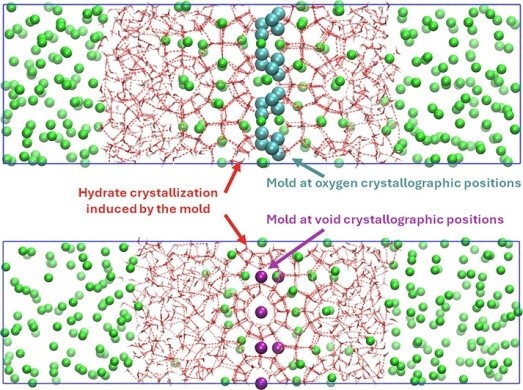
Calculations: In this work, we present a direct molecular simulation of the hydrate–water interfacial free energy using two novel and independent extensions of the mold integration method. These extensions are specifically designed to induce the formation of a thin, planar hydrate–water interface and to compute the reversible work required to create it. For this purpose, we employ the TIP4P/Ice force field—one of the most reliable water models available—known for accurately reproducing the melting temperature of ice Ih under ambient conditions.
Findigns: Our results show that the interfacial free energy of hydrate is significantly higher than that of hydrate, offering a natural explanation for their markedly different nucleation behaviors. This aligns with prior predictions based on Classical Nucleation Theory, as well as advanced sampling techniques such as Transition Path Sampling and Transition Interface Sampling. Notably, our computed value for hydrate approaches ~ 40 mJ/m2, in agreement with the upper bound of existing experimental estimates, while values for hydrate and ice Ih remain closer to ~ 30 mJ/m2 . This direct, theory-independent determination provides new insights into the molecular mechanisms underlying hydrate formation and offers robust benchmarks for predictive modeling and the design of hydrate-based technologies.