S. Dorado-Alfaro, E. Hernández, J. Algaba, P. Navarro, F. J. Blas, J. Palomar, J. Mol. Liquids, 2025
https://doi.org/10.1016/j.molliq.2025.127322
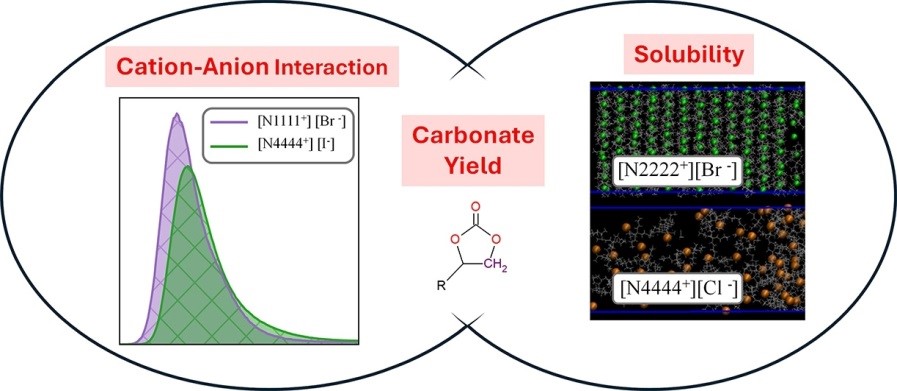
Climate emergency has led to the investigation of CO2 valorization routes. A competitive process included in this framework is the catalytic CO2 cycloaddition to epoxides, to produce cyclic carbonates. Halide-based Ionic liquids (ILs) have been postulated to be a competitive choice. Nevertheless, the structure-performance relation for different ILs is still a topic of debate, being the cation-anion dissociation constant a key descriptor. In this work, the ions effect is tackled by Molecular Dynamics (MD) simulations. Propylene oxide and carbonate force fields were tested and used for 1,2-epoxyhexane and hexylene carbonate force field construction, while ILs were modelled by the CL&P force field. Solubilities in an epoxide-carbonate medium were tested for ILs composed of [N4444+] or [N2222+] cations combined with the halide anions: Iodide [I−], Bromide [Br−] and Chloride [Cl−]. Results showed that [N2222+] cation-based ionic liquids were insoluble in the epoxide/carbonate medium, whereas [N4444+] cation-based ionic liquids demonstrated diffusion. Reaction medium interactions were studied between key atoms for experimentally soluble ILs. It was found that cation-anion interaction follows the catalytic activity trend, being [I−] the halide anion less associated with ([N4444+], [bmim+] and [emim+]) cations. A correlation between the first peak integration of the radial distribution functions and the experimental yields (including [N1111+][Br−]) could be established with a regression coefficient of 0.86. Additionally, [I−] based ILs displayed a better interaction between the cation and the epoxide oxygen, phenomena linked to epoxide activation and intermediates stabilization. Therefore, the path towards the understanding of this catalytic system has been widened.