H. Videa, K. M. Byrne, T. Krämer, A. J. Martínez-Martínez. Angew. Chem. Int. Ed., 2026
https://doi.org/10.1002/anie.7183298
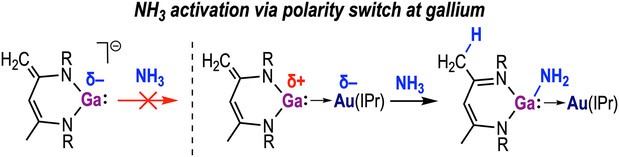
Controlling the reactivity of low-valent main-group nucleophiles in small-molecule activation remains a significant challenge. Here, we report the synthesis of a family of charge-delocalized β-diketiminate gallyl anions, accessible across the Li–Cs series, whose aggregation and ion pairing can be tuned to furnish a cation-gated gallium(I) nucleophilic platform. Only the cesium crown-encapsulated derivative forms a truly separated ion pair (SIP) that exhibits clean nucleophilic reactivity toward (IPr)AuCl to generate an unsupported Ga─Au bimetallic complex, isolable, and structurally authenticated. Coordination at Au enhances the Lewis acidity at gallium(I), enabling cooperative N─H and O─H σ-bond activation under mild conditions via a substrate-assisted proton-shuttle pathway. This reactivity, exemplified by NH3 and H2O, and extended to MeNH2 and MeOH, is inaccessible to the free gallyl anions. These findings define a new gallium-based nucleophilic platform for exploiting bimetallic cooperation in small-molecule bond activation.