J. Martínez-Laguna, I. L. Schmidtke, A. Monti, R. J. Rama, D. Sánchez-Rodas, M. Nicasio, A. Nova, A. Caballero, P. J. Pérez. Chem., 2026
https://doi.org/10.1016/j.chempr.2026.102999
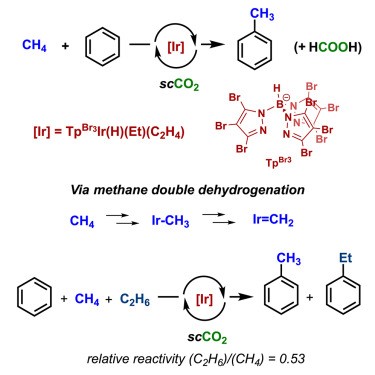
During our investigations to develop homogeneous catalysts for methane functionalization, we have found that methane can be dehydrogenated under homogeneous conditions to form Ir=CH2 units that can react further with benzene, producing toluene. This transformation adds to the limited scope of metal-catalyzed methane functionalization reactions. The same method can be applied to ethane, producing ethylbenzene. These transformations proceed via methylidene or ethylidene intermediates (Ir=CHR, R = H or Me), formed through a double dehydrogenation of methane or ethane, respectively. Experimental and theoretical investigations, including density functional theory analysis and microkinetic modeling, support the proposed mechanism. The discovery of methane dehydrogenation with iridium centers and the formation of methylidene (Ir=CH2) intermediates in homogeneous C–C coupling reactions could open novel avenues for the formation of hydrocarbons, eliminating the need for syngas required in the Fischer-Tropsch process.