C. Romero-Guzmán, J. Algaba, M. M. Conde, M. M. Piñeiro, F. J. Blas. J. Chem. Phys., 2026
https://doi.org/10.1063/5.0325832
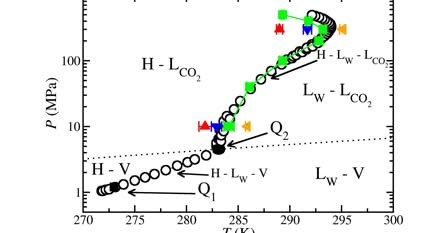
Over the past decade, the phase equilibria of CO2 hydrates have been the subject of numerous molecular simulation studies. However, inconsistencies among these works—stemming from disparate molecular models, box geometries, and simulation protocols—have hindered direct comparison. Moreover, prior simulations have generally not reported the three-phase dissociation temperature, T3, under conditions that reliably suppress finite-size and intermolecular potential truncation artifacts, often because of limited system sizes and/or short cutoff distances. Here, we compute T3 using molecular simulations specifically designed to minimize these effects, employing systems containing more than 3000 molecules and a long cutoff of 1.6 nm. The hydrate–aqueous–liquid CO2 coexistence temperature is obtained via the solubility method: at fixed pressure, we evaluate the CO2 solubility in the aqueous phase in contact with a CO2 hydrate phase and, separately, with a pure liquid CO2 phase over a range of temperatures. The intersection of the two solubility curves defines T3. This approach has emerged in recent years as an alternative and complementary method to direct coexistence. We apply this procedure from 100 to 5000 bar. In addition, we refine the unlike CO2–water interactions to reproduce the dissociation line quantitatively. We find that modifying the Berthelot combining rule for CO2–water cross-interactions with a scaling factor ξ = 1.085 yields excellent agreement between simulated T3 values and experimental ones, providing a qualitative description of the dissociation enthalpy along the line. Furthermore, we provide, to the best of our knowledge, the first physical explanation for the unusual reentrant behavior of the CO2 hydrate dissociation line. We show that this behavior originates from a pressure-induced change in the sign of the reaction molar volume, caused primarily by the strong compressibility of the fluid CO2 phase relative to the hydrate and aqueous phases.