G. Rigoni, M. Casciotti, T. R. Belderrain, P. J. Pérez, M. Albrecht, Organometallics, 2024
https://doi.org/10.1021/acs.organomet.4c00398
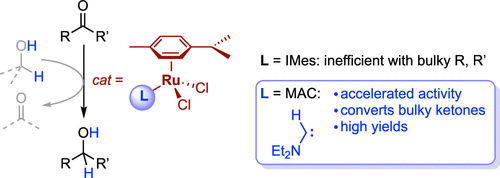
While catalytic transfer hydrogenation of ketones is well established for model substrates, major limitations are faced when applying this methodology to sterically encumbered carbonyl substrates. To address this challenge, we have here combined the catalytic competence of ruthenium together with a unique monosubstituted amino-carbene (MAC) as a ligand to produce a ruthenium complex, Ru-MAC, which features a markedly reduced buried volume. We demonstrate that Ru-MAC is a transfer hydrogenation catalyst with outstanding activity when compared to other related ruthenium complexes such as Ru-IMes, the analogue containing the Arduengo-type N,N′-mesityl substituted imidazolylidene ligand. The catalytic acceleration factor of Ru-MAC is particularly pronounced with sterically bulky ketones. Multivariate regression analysis allowed us to deduce a model that quantitatively correlates the steric parameters of the substrates with the catalytic acceleration factor, revealing paramount relevance of the minimum width of the larger ketone substituent, viz. Sterimol parameter B1L. The exquisite activity of Ru-MAC is further demonstrated by the transfer hydrogenation, for the first time, of two extremely hindered substrates, namely 2,4,6-triisopropylacetophenone and pentamethylacetophenone, to quantitatively yield the corresponding alcohols.